United States Patent5,854,404
Nanba , et al.December 29, 1998
Antitumor substance extracted from grifola

Abstract
An antitumor substance having high immunopotentiating activity, extracted and fractionated from mycelia or fruit bodies of Grifola with water can be obtained by adding alcohol to its extract at a final concentration of 20 to 60%, preferably at a final concentration of 20 to 50% by volume (low-concentration addition) to remove floating or adhering matter from it.
Inventors:Nanba; Hiroaki (Hyogo, JP), Kubo; Keiko (Hyogo, JP)
Assignee:Yukiguni Maitake Co., Ltd. (Niigata, JP)
Family ID:12899202
Appl. No.:08/812,795
Filed:March 6, 1997
Foreign Application Priority Data
Mar 8, 1996 [JP]8-051880
Current U.S. Class:530/424; 530/300; 530/418; 530/412; 530/350
Current CPC Class:A61K 36/07 (20130101); C07K 14/375 (20130101); A61P 37/04 (20180101); A61P 35/00 (20180101)
Current International Class: C07K 14/37 (20060101); C07K 14/375 (20060101); C07K 001/00 ()
Field of Search: 514/12 ;530/412,414,418,424,300,350
Other References
Nanba et al Chem Pharm Bull. vol. 35 (3) 1162-1168, 1987..
Primary Examiner: Huff; Sheela
Assistant Examiner: Reeves; Julie E.
Attorney, Agent or Firm: Davidson, Davidson & Kappel, LLC
Claims
What is claimed is:
1. A glucan/protein complex produced by the steps of:
(a) thermally extracting mycelia or fruit bodies of Grifola with water at a temperature of from 50.degree. C. to 135.degree. C.;
(b) adding alcohol to the resulting water-soluble extract at a final concentration of 20 to 60% by volume, allowing said extract to stand in a vessel at a temperature of 1.degree. to 25.degree. C., and removing floating matter on the liquid or in the liquid or matter adhered to the vessel wall; and
(c) addition alcohol to said extract at a final concentration of 80 to 99% by volume, allowing said extract to stand at 1.degree. to 25.degree. C., and recovering said glucan/protein complex in the form of precipitates.
2. A glucan/protein complex produced by the steps of:
(a) thermally extracting mycelia or fruit bodies of Grifola with water at a temperature of from 50.degree. C. to 135.degree. C.;
(b) adding alcohol to the resulting water-soluble extract at a final concentration of 20 to 50% by volume, allowing said extract to stand in a vessel at a temperature of 1.degree. to 5.degree. C., and removing floating matter on the liquid or in the liquid or matter adhered to the vessel wall; and
(c) adding alcohol to said extract at a final concentration of 80 to 90% by volume, allowing said extract to stand at 1.degree. to 5.degree. C., and recovering said glucan/protein complex in the form of precipitates.
3. A glucan/protein complex produced by the steps of:
(a) thermally extracting mycelia or fruit bodies of Grifola with water at a temperature of from 50.degree. C. to 135.degree. C.;
(b) adding alcohol to the resulting water-soluble extract at a final concentration of 20 to 60% by volume, allowing said extract to stand in a vessel at a temperature of 1.degree. to 25.degree. C., and removing floating matter on the liquid or in the liquid or matter adhered to the vessel wall; and
(c) concentrating said extract to form said glucan/protein complex in the form of precipitates, or concentrating said extract into dryness to form said glucan/protein complex.
4. A glucan/protein complex produced by the steps of:
(a) thermally extracting mycelia or fruit bodies of Grifola with water at a temperature of from 50.degree. C. to 135.degree. C.;
(b) adding alcohol to the resulting water-soluble extract at a final concentration of 20 to 50% by volume, allowing said extract to stand in a vessel at a temperature of 1.degree. to 5.degree. C., and removing floating matter on the liquid or in the liquid or matter adhered to the vessel wall; and
(c) concentrating the solution to form said glucan/protein complex in the form of precipitates, or concentrating said extract into dryness to form said glucan/protein complex.
5. The glucan/protein complex according to any one of claims 1, 2, 3 or 4, wherein the ratio of glucan to protein is in the range of from 80:20 to 99:1.
6. An antitumor agent having tumor-growth inhibition and cellular immunocompetent activity, which consists essentially of the glucan/protein complex of any one of claims 1, 2, 3 or 4 as an active ingredient.
7. The glucan/protein complex according to any one of claims 1, 2, 3 or 4, wherein Grifola is Maitake, Grifola albicans Imaz, Grifola umbellatus or Grifola gigantea.
8. The glucan/protein complex according to claim 2, wherein the ratio of glucan to protein is in the range of from 80:20 to 99:1.
9. The glucan/protein complex according to claim 3, wherein the ratio of glucan to protein is in the range of from 80:20 to 99:1.
10. The glucan/protein complex according to claim 4, wherein the ratio of glucan to protein is in the range of from 80:20 to 99:1.
11. An antitumor agent having tumor-growth inhibition and cellular immunocompetent activity, which consists essentially of the glucan/protein complex of claim 2 as an active ingredient.
12. An antitumor agent having tumor-growth inhibition and cellular immunocompetent activity, which consists essentially of the glucan/protein complex of claim 3 as an active ingredient.
13. An antitumor agent having tumor-growth inhibition and cellular immunocompetent activity, which consists essentially of the glucan/protein complex of claim 4 as an active ingredient.
14. An antitumor agent having tumor-growth inhibition and cellular immunocompetent activity, which consists essentially of the glucan/protein complex of claim 5 as an active ingredient.
15. The glucan/protein complex according to claim 2, wherein Grifola is Maitake, Grifola albicans Imaz, Grifola umbellatus or Grifola gigantea.
16. The glucan/protein complex according to claim 3, wherein Grifola is Maitake, Grifola albicans Imaz, Grifola umbellatus or Grifola gigantea.
17. The glucan/protein complex according to claim 4, wherein Grifola is Maitake, Grifola albicans Imaz, Grifola umbellatus or Grifola gigantea.
Description
FIELD OF THE INVENTION
The present invention relates to an antitumor substance having high immunopotentiating activity, which was extracted and fractionated from mycelia or fruit bodies of a "Maitake" mushroom (Grifola).
BACKGROUND OF THE INVENTION
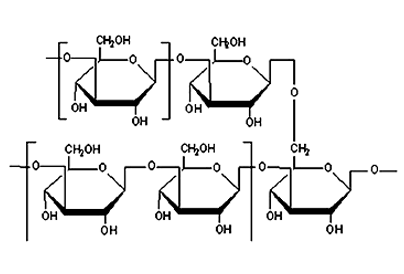
Polysaccharides consisting of .beta.-1,6-linked glucose main chain with .beta.-1,3-linked glucose branches or consisting of .beta.-1,3-linked glucose main chain with .beta.-1,6-linked glucose branches extracted from mycelia or fruit bodies of Grifola, is known to have anticancer activity (see Japanese Patent LOP Publication No. 210901/1984).
A process for producing an anticancer substance, which comprises a combination of the steps of extracting Grifola, Grifola gigantea (Tonbimai) or Laetiporus sulphureus (Masutake) with hot water, concentrating the extract under reduced pressure, precipitating the concentrate with an organic solvent, dialyzing the precipitates to remove low-molecular-weight substances, and extracting impurities with a lipophilic organic solvent to remove them from the Jialysate, is also known (see Japanese Patent Publication No. 16047/1968).
However, the prior processes described in Japanese Patent LOP Publication No. 210901/1984 and Japanese Patent Publication No. 16047/1968 are not necessarily appropriate for providing a larger amount of pharmaceutical preparations and health foods efficiently from limited resources because their purification steps are considerably complicated and the products contain substances inhibiting immunopotentiating activity.
EFFECT OF THE INVENTION
The above results indicate that immunopotentiating activity and tumor growth inhibitory activity are enhanced by removing the floating matter on the liquid or in the liquid or the adhering matter to the vessel wall occurring by adding alcohol at a final concentration of 20 to 60%, preferably at a final concentration of 20 to 50% by volume to the hot water extract from Grifola.
Hence, the feature of the present invention lies not in simply extracting polymeric .beta.-glucan, but in effectively providing a glucan/protein complex having high immunopotentiating activity from limited resources by a simple method.
The substance obtained according to the present invention is of low toxicity and high safety and can be orally administered as health foods and pharmaceutical preparations, especially antitumor agent, in the form of tablets, capsules, liquid, syrup etc.